All about genetics
20 mins read
This advice applies across the UK.
This section is written for the non-specialist individual, concentrating on two forms of inheritance, the single abnormal gene which is changed, or ‘mutated’, and chromosomal abnormalities.
In this article
Many traits in plants and animals are heritable (can be passed down from one generation to the next), and genetics is the study of these heritable factors. Specific conditions and rare syndromes may have a genetic basis. Where this is the case, there will be a variety of causes. For example, the causes may include a single abnormal gene, a chromosomal abnormality or a genetic predisposition, meaning a person has a higher chance compared with the general population, to have a particular disease or condition.
This section is written for the non-specialist individual. We shall concentrate on two forms of inheritance, the single abnormal gene which is changed, or ‘mutated’, and chromosomal abnormalities.
The human body is made up of billions of cells. At the centre of each cell is a special compartment called the nucleus, which stores threads of deoxyribonucleic acid (DNA). These threads are wrapped in structures called chromosomes. Chromosomes are composed of around 50,000 genes. Genes are composed of small stretches of DNA and are our body’s genetic ‘blue print’, giving important instructions to build all the different molecules our bodies need to carry out important chemical reactions and processes that keep us alive. Genes are also responsible for determining normal traits such as blood groups or hair colour, as well as causing disease when the genes are damaged or mutated.
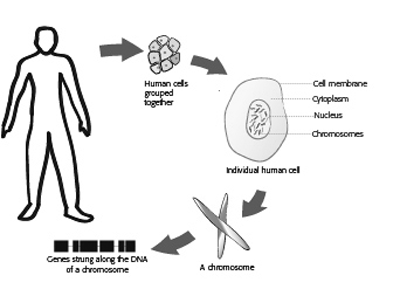
Apart from the sex cells: the ovum (the ‘egg’ from the female) and the sperm (from the male), every cell in the human body normally contains 23 pairs of chromosomes (46 in total). This number of chromosomes is known as the ‘diploid’ number. Of the 23 pairs of chromosomes in each cell, one pair is from the mother and one from the father. The first 22 of our pairs of chromosomes are called autosomal chromosomes. The chromosomes in the twenty-third pair are called the sex chromosomes (X and Y), named because they determine the sex of an individual. Males have an X and Y chromosome, while females have two X chromosomes.
The chromosomes in our sex cells undergo a special process of division, known as meiosis so that they contain 23 ‘single’ human chromosomes (instead of 23 pairs). This ‘half number’ of chromosomes is known as the ‘haploid’ number. In this way, the mother contributes half the genetic material and the father contributes half of the genetic material of a child.
When a baby is conceived, one ovum and one sperm fuse together. Upon fertilisation, there are a total of 23 pairs of chromosomes, making 46 chromosomes in total. Inheritance will depend upon the arrangement of the genes on our chromosomes and the way in which genes act when they are passed down through generations.
Single abnormal gene
An abnormal or mutated gene may be considered as a variant of a ‘normal’ gene because it is changed in some way and therefore does not give out the instruction that it should do. This change may occur spontaneously by chance and have no significance for the individual concerned.
In other cases, a gene that mutates (changes its character) may give rise to specific inherited disorders where there is no previous family history. Inheritance of the mutated gene may be autosomal dominant, autosomal recessive or X-linked recessive (see Types of inheritance).
Research has shown that, in many conditions, there may be different types of mutation in a single gene, all of which can result in the same clinical outcome. For example, in cystic fibrosis over two hundred different mutations can occur in the implicated gene, but they mostly produce the same disease pattern and symptoms in individuals with cystic fibrosis.
Types of inheritance
Autosomal inheritance means that males or females are equally affected, this is because the mutation is not passed on the sex chromosomes (the X or Y chromosomes) and, therefore, there is not a higher chance of either a male or female receiving the mutation.
Autosomal dominant inheritance
Some conditions are caused by a mutation in a gene that is dominant. As a person receives two copies of every gene (one from the mother and one from the father), the dominant mutated gene overrides the normal gene and the individual will be affected by that genetic condition. In dominant inheritance, the chance of passing on the disorder is 50 per cent for each pregnancy, (as illustrated in the diagram below).
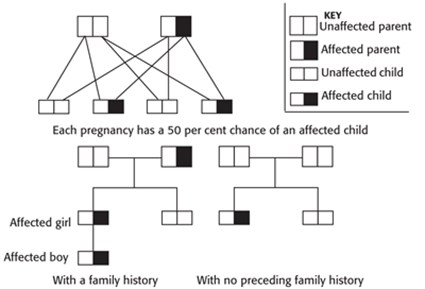
Examples of conditions that are inherited in an autosomal dominant manner are Huntington’s disease or Tuberous Sclerosis Complex.
In some cases, there is some variability in the expression of the dominant gene, which is known as ‘penetrance’. Penetrance is not complete in some individuals, resulting in a milder form of the condition rather than the full effect usually seen when inheriting the dominant gene.
Sometimes a condition with autosomal dominant inheritance may arise due to a mutation in ovum or sperm that joined to make a baby. When this happens and the parent of the child is not affected (there is no preceding history), the process is known as a ‘sporadic’ mutation. Sporadic mutations can arise for a number of reasons, but are usually caused by an error in copying the genetic material. It is unlikely for parents to have another child affected by the same sporadic mutation. The occurrence of a sporadic mutation is unlikely to be as a result of anything the parents have or have not done.
Autosomal recessive inheritance
In this form of inheritance, the affected gene is recessive. This means that a person must inherit two copies of the mutated gene, one copy from each parent, to be affected by the disorder. If a person inherits just one copy of a mutated gene and one normal copy then, in most cases, the person will not be affected by the condition but is a healthy ‘carrier’. Being a carrier means that a person does not have the condition but carries a mutated copy of the gene, which can be passed on to future generations.
Each child of parents that both carry the same mutated gene therefore has a 25 per cent chance of inheriting a mutated gene from both parents and, therefore, being affected by the condition. This chance remains the same for all pregnancies and is the same for males and females.
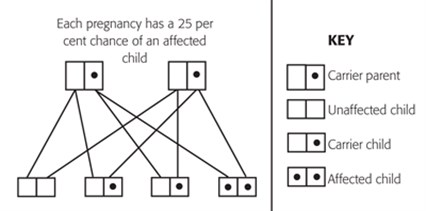
There is also a 50 per cent chance that the child will inherit just one mutated copy of the gene from a parent. In this case the child will be a healthy carrier like their parent.
Examples of conditions that are inherited in an autosomal recessive manner are Friedreich’s Ataxia, Cystic Fibrosis or Phenylketonuria.
Unless the parents are related, the chances of marrying a carrier with the same mutated recessive gene are low. The incidence of recessive genes in the population varies with different conditions. A number of testing options may be available for people who have a family history of a recessive genetic condition and this information may be useful when planning pregnancies. Genetic counselling or advice should be sought if there is a family history of a recessive genetic condition.
X-linked inheritance
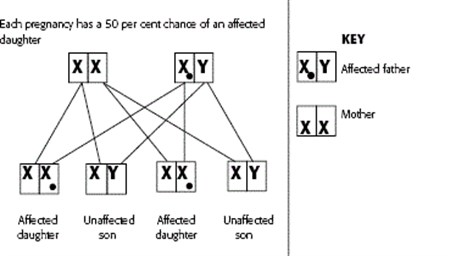
As discussed earlier, one pair of chromosomes determines a person’s sex, with a female having two X chromosomes and a male having one X and one Y chromosome. Females inherit one X chromosome from their father and one X chromosome from their mother, whereas males inherit an X chromosome from their mother and their Y chromosome from their father. The X chromosome has many genes, whereas the Y chromosome is smaller and contains fewer genes. Sometimes a mutation occurs on the X chromosome and genetic conditions that occur as a result of this kind of mutation are known as X-linked genetic conditions.
X-linked recessive inheritance
This is a recessive form of inheritance where the mother carries the mutated gene on the X chromosome. As females have two copies of the X chromosome, if there is a mutated gene on one of their copies the normal copy on the other X chromosome compensates for the mutated gene. In this case, the female is usually unaffected by the condition and is a carrier. As a male only has one copy of the X chromosome, if this has a mutated gene they will be affected with the condition that this causes. In some cases, females may display mild symptoms of the condition. An example of this is Fragile X Syndrome. Usually though the female is less affected than male counterparts.
Examples of conditions inherited in an X-linked recessive manner include Duchenne Muscular Dystrophy, Haemophilia or Hunter disease (a mucopolysaccharide disease, see entry Mucopolysaccharide diseases and Associated diseases).
If a female carrier of an X-linked recessive condition has a daughter, she will pass on either the X chromosome with the normal gene or the X chromosome with the mutated gene. Each daughter of a female carrier, therefore, has a 50 per cent chance of inheriting the mutated gene and being a carrier like their mother. If the daughter does not inherit the mutated gene, she is not a carrier.
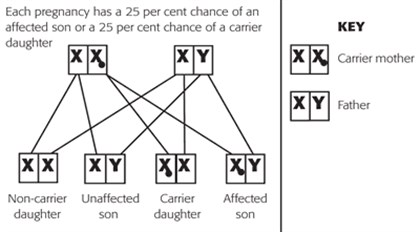
If a male with an X-linked condition has a daughter, he will always pass on one copy of the mutated gene to his daughters and they will be carriers. This is because males only have one X chromosome.
If a male with an X-linked condition has a son, then his son will never inherit the mutated gene on the X chromosome. This is because men always pass their Y chromosome to their sons.
X-lined dominant inheritance
Very rarely, X-linked conditions are passed on in a dominant way, one such example is Coffin-Lowry syndrome. This means that even though a female inherits one normal copy of the gene and one mutated copy, she will have the condition as the dominant mutated gene overrides the normal copy. If a male inherits a mutated gene on the X chromosome then he will have the condition, as he will only have one copy of the X chromosome.
An affected female will have a 50 per cent chance of passing the disorder on to both her sons and her daughters. An affected male will pass the condition on to all his daughters, but not to his sons.
Mitochondrial inheritance
The DNA in a cell is largely located in the nucleus of the cell, but in the surrounding cytoplasm (jelly-like material that holds together cell components) of the cell there are small bodies called mitochondria, which are responsible for energy production. Mitochondria carry their own genes and DNA. These genes can also be passed on during reproduction. However, the pattern of inheritance is not always predictable since there is a chance element in determining the amount of cytoplasm and hence the amount of mitochondrial DNA that is passed on.
Mitochondrial DNA is passed on through the egg but not by the sperm as it is only the nucleus of the sperm that enters the egg during fertilisation. Therefore, the pattern we see with mitochondrial inheritance is transmission through an affected female to a variable number of male and female offspring, but no transmission from an affected male.
Imprinted genes
In most cases, it will not matter whether the gene or chromosome defect is inherited from the mother or father – the effect on the child will be the same. However, there are some mutations in genes and chromosome in which there will be a different effect depending on which parent the abnormality has come from. For example, a deletion of chromosome 15 from the father in the sperm will cause Prader-Willi syndrome, whereas the same deletion of chromosome 15 from the mother in the ovum (egg) will cause a different condition called Angelman syndrome.
With these imprinted genes, it is necessary to have both the maternal (that of the mother) and paternal (that of the father) contribution in early embryonic development in the womb. It is likely that the need for a contribution from both parents has arisen in evolutionary terms with sexual reproduction.
Chromosomal abnormalities
A chromosome is a rod-like structure present in the nucleus of all cells within the body, with the exception of the red blood cells. A chromosome has a centromere in the centre from which the arms radiate. The q arms are long arms and the p arms are the short arms.
A chromosomal change or abnormality occurs when there is a defect in a chromosome or in the arrangement of the genetic material on the chromosome. Chromosomal abnormalities give rise to specific physical features, but it should be stressed that there may be wide variations in the severity of the symptoms in individuals with the same chromosome abnormality.
Additional material may be attached to a chromosome, absence of a whole or part of a chromosome may occur and defective formation of the chromosome may also occur. Increases and decreases in chromosomal material, and therefore genes, interfere with normal body function and development.
There are two main types of chromosomal abnormality that may occur during meiosis (specialised sex cell division) and fertilisation. These are known as numerical aberrations (changes in the number of chromosomes from the norm) and structural aberrations (where the normal structure of a chromosome has changed).
Numerical aberrations
Sometimes there is a failure in chromosome division (meiosis) in the ovum and sperm before they fuse and there maybe extra or fewer chromosomes. This can result in anomalies such as Down’s syndrome (47 chromosomes, with three copies of chromosome 21) or Turner syndrome (45 chromosomes where the second X chromosome in females is missing or abnormal). The following are examples of numerical aberrations:
- triploidy – this is where a cell has one extra set of chromosomes, so there are 69 chromosomes instead of 46
- mosaicism – this is where only some of the body’s cells carry an extra set of chromosomes
- trisomy – this is where one extra complete chromosome is present, so the number of chromosomes in each affected cell would be 47 instead of 46
- monosomy – this is when a complete chromosome is missing and the number of chromosomes in a cell is 45 instead of 46.
Structural aberrations
These occur where there is a rearrangement in the location of, or a loss of, genetic material. They occur because of breakages in a chromosome, which can either be ‘de novo’ occurring spontaneously, or be inherited from a parent. Structural disorders include the following:
- insertion – this occurs when a segment in one chromosome inserts into a gap in another chromosome
- deletion – this involves a loss of a part or segment of a chromosome. Very small deletions are known as microdeletions
- duplication – this occurs when an extra copy of a segment of a chromosome is present. These are sometimes known as partial trisomy. If a person has two extra copies of a chromosomal segment, then this is known as a triplication or a partial tetrasomy. Very small duplications are known as microduplications
- inversions – this occurs when there are two breaks in a single chromosome. The segment between break points rotates 180° and reinserts itself back into the gap created by the original breaks
- ring formations – this occurs when the ends of both arms of the same chromosome are deleted and the remaining broken ends become ‘sticky’ and join together to make a ring shape, in these cases it is the deleted DNA that is significant. Sometimes ring chromosomes are extra chromosomes and in which case it is the extra material that is significant and causes symptoms in a person
- reciprocal translocation (balanced, unbalanced and Robertsonian) – this occurs when DNA is transferred from one non-homologous (as in not in a pair with the other chromosome of the same number) chromosome to another. In a balanced translocation, there are breaks in two or more chromosomes and the resulting DNA fragments swap places. If genetic material is lost then the translocation is known as ‘unbalanced’. Robertsonian translocations occur when the short arms of chromosomes 13, 14, 15, 21 or 22 are lost and the remaining long arms fuse together.
Identifying disease-causing genes
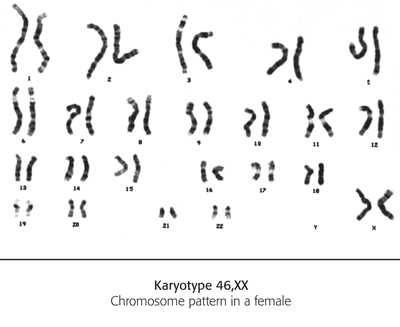
Karotyping
This is the analysis of chromosomes under a light microscope. The test can be performed on almost any tissue, including amniotic fluid (fluid taken in an amniocentesis test), blood and bone marrow. Chromosomes are stained and then photographed to show the arrangement of the chromosomes. Certain abnormalities can be identified through the number or arrangement of the chromosomes. These can include large deletions and extra chromosomes.
Fluorescent in Situ Hybridisation (FISH)
In Fluorescent in Situ Hybridisation (FISH) known segments of DNA (called probes) are fluorescently labelled and used to analyse chromosomes. The probes are mixed with samples from the person being tested containing their chromosomes, often taken from blood samples, and bind to small parts of a chromosome. Once bound with the chromosomes the probes mark them with fluorescent colours, which can be visualised under a fluorescent microscope. If a probe for an area of DNA which is known to cause a genetic condition if deleted is not present on FISH, it can be assumed that this area of DNA is missing. FISH is useful if the chromosome suspected to have a change in it is known.
Microarray comparative hybridisation (array-CGH)
This is a new technique and allows identification of small changes in genetic material that it may not be possible to identify with karotyping. Unlike FISH the chromosome on which the change is suspected to be on does not need to be known and a person’s whole genome (all their DNA) can be checked for changes that may cause a genetic condition.
This technique compares reference DNA to that of the patient, allowing the test to distinguish differences between the two sets of DNA. In this way, deletions or duplications and the affected genes can be identified. The patient and reference DNA are labelled with different colour fluorescent dyes and attached to a special glass slide called a microarray. The DNA is allowed to bind together sticking to sequences that match exactly and the fluorescence emitted is measured. If there is a lot more fluorescence from the reference DNA compared with the patient’s DNA for one probe, this means that some DNA is missing in the patient (eg deletion) and vice versa.
Descriptions of particular chromosomal formations are often written in a shortened form. This indicates the total number of chromosomes, the sex of the individual and the abnormal chromosome number.
For example, a girl with Cri du Chat syndrome would be shortened to 46,XX,5p – that is, the affected child has 46 chromosomes, is a female (XX) and has a deletion (indicated by the minus sign) of the short arm of chromosome 5 (which is named the p arm). If this chromosome was in the form of a ring it would be 46,XX,r(5), the ‘r’ standing for ring. A trisomy could be written as 47,XY, + 21, for a male with Down’s syndrome, indicating an additional copy of chromosome 21.
Genetic pre-disposition in multifactorial disorders
Conditions such as isolated malformations or common diseases like diabetes or heart disease combine a genetic pre-disposition to develop a disease with other environmental factors that may also contribute. There may be an undefined family history of the condition. Where two affected children are born in the same family, there may be an increased risk of recurrence. An example of a condition which falls into this category is cleft lip and/or palate.
Genetic counselling
Genetic counselling can usually calculate risks of parents having a child affected by a genetic condition. It can also provide support and advice for families who already have an affected child and wish to enlarge their family. A list of regional genetics centres are provided by the Genetic Alliance UK.
Prenatal diagnosis
There are a number of techniques that are used to diagnose conditions in unborn babies whose mothers are at risk of having a baby with an abnormality. Risks may include a family history of an abnormality, or that the parents have already had one child with, for example, a heart defect. On the other hand, prenatal testing may be performed on the grounds of the age of the mother.More information about screening in pregnancy is available on the NHS website.
Ultrasound scanning
This technique involves the use of ultrasonic waves (sound waves of a high frequency which cannot be heard by the human ear) to scan the unborn baby (fetus) and measure it. The fetus can be seen on a computer screen attached to the scanner enabling bone problems and other abnormalities to be identified. Fetal measurements taken at the scan can be compared with average ‘normal’ fetal age measurements to identify abnormalities.
Amniocentesis
The amniotic sac is the bag of fluid in which the baby floats in the womb. Amniocentesis is a way of removing some of this fluid for further analysis by passing a fine needle through the abdomen (belly area) into the womb. The sample of fluid is then analysed and certain biochemical, chromosomal or neural tube defects can be identified. Amniocentesis can identify metabolic diseases (where the affected enzyme has been previously identified) and chromosome defects; is usually offered at around 14 weeks of pregnancy. There is a very small chance (around 0.5 per cent) of miscarriage after amniocentesis.
Chorionic villus sampling
After fertilisation of the ovum by the sperm, a cell mass is formed. The inner cells of this mass form the fetus, while the outer cells become embedded in the wall of womb forming the placenta (a connection between the mother and baby through which oxygen and nutrients pass). These placenta cells are called chorion cells and can be removed for analysis in a test called chorionic villus sampling or (CVS).
The cells are removed using a fine needle that passes through the abdominal wall and aspirating (drawing) some tissue back into a syringe. This test can be performed at 11 to 13 weeks and can identify metabolic defects where the affected enzyme has been isolated, chromosomal defects and certain single gene defects where the specific gene has previously been identified. There is a very small chance (around one per cent) of miscarriage after CVS.
Related information
Further information on genetics, patterns of inheritance and chromosome disorders can be found by accessing the following resources.
Genetics Homepage Reference
- Easy-to-understand information on genes, DNA and genetic conditions http://ghr.nlm.nih.gov/BrowseGenes
The Genetic Alliance UK
- Easy-to-understand introduction to genetics and genetic conditions
http://www.geneticalliance.org.uk/education1.htm
- A range of information for patients on modes of inheritance and genetic testing
http://www.geneticalliance.org.uk/education2.htm
- The Genetic Alliance also has listings of all the regional genetic testing centres in the UK
http://www.geneticalliance.org.uk/services.htm
Unique – the rare chromosome disorders support group
- Information on DNA, genes and inheritance can be found in their guide Little yellow book: Vol 1: A guide to rare chromosome disorders
http://www.rarechromo.org/html/LittleYellowBook.asp
- Unique also have a range of information leaflets for specific chromosome disorders
Medical text written June 1991 by Professor Michael Patton. Last updated November 2010 by Professor Michael Patton, Professor of Medical Genetics, St George’s Hospital Medical School, London, UK.